HS Dosing COSENTYX® (secukinumab)
€ 12.00 · 5 (101) · En stock


Novartis' Cosentyx clinches FDA nod in lesser-known skin disease

Secukinumab Gains FDA Approval to Treat Hidradenitis Suppurativa

Full article: Real-world dose escalation of biologics for moderate-to-severe psoriasis in the United States

Cosentyx - NPS MedicineWise

Cosentyx Dosage Guide
COSENTYX® (secukinumab) for US HCP

Impact of Secukinumab on Endothelial Dysfunction and Other Cardiovascular Disease Parameters in Psoriasis Patients over 52 Weeks - ScienceDirect

Pharmaceutics, Free Full-Text
COSENTYX® (secukinumab) for US HCP

WO2019097493A1 - Treating hidradenitis suppurativa with il-17 antagonists - Google Patents
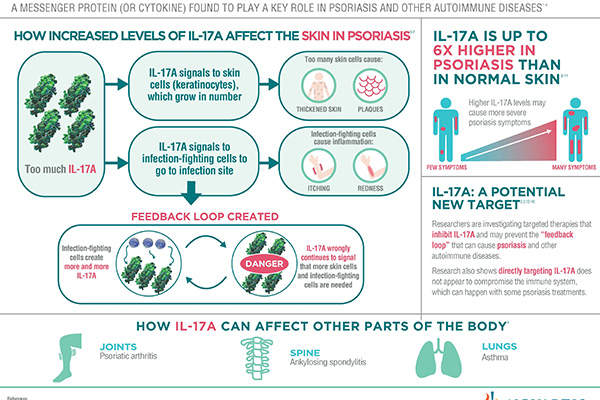
Cosentyx (secukinumab) for Treatment of Plaque Psoriasis - Clinical Trials Arena
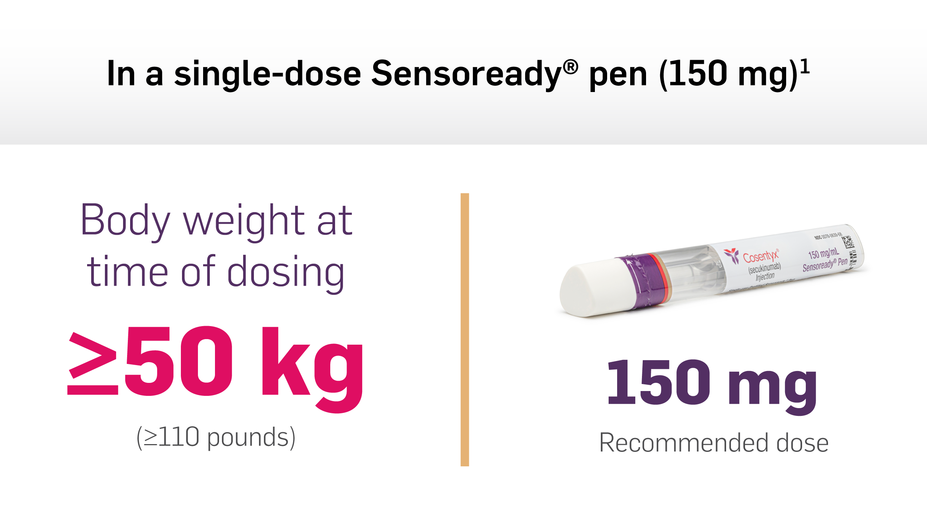
Dosing & Devices, COSENTYX® (secukinumab)
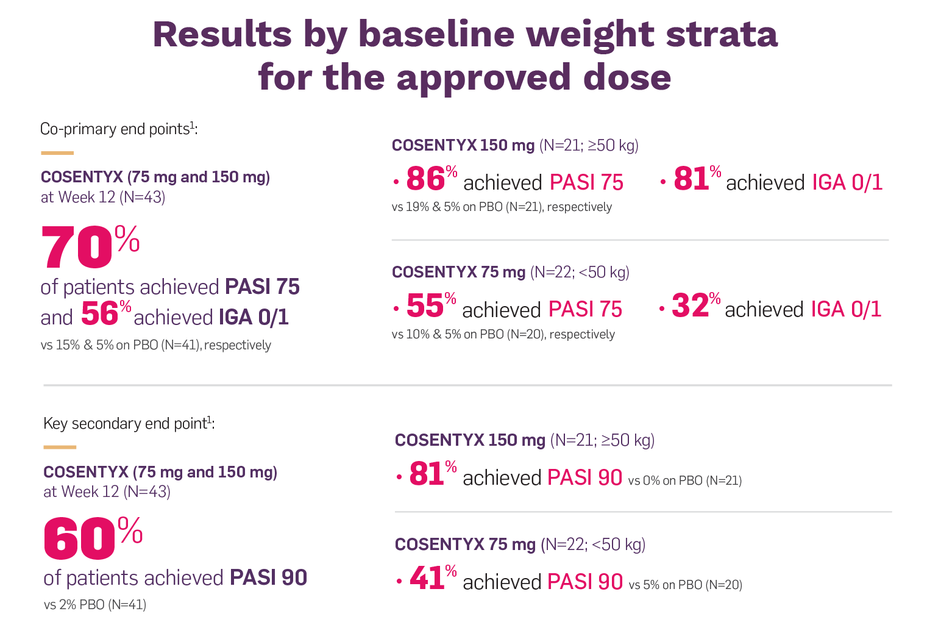
Pediatric PsO Treatment Information, COSENTYX® (secukinumab)